Provaxa
Your Strategic & Trusted Medical Research Partner
We bring together a single window and powerful network of services offering integrated solutions for sponsors seeking speed, compliance, and innovation in the global markets.
With a shared commitment to ICH-GCP, ISO-certified quality systems, and regulatory excellence, we accelerate pathways from concept to cure.
Who We Are
A diversified healthcare enterprise transforming scientific innovations into tangible patient outcomes
We are a diversified healthcare enterprise with strong foundations in preclinical and clinical research, stem cell and gene therapy, biopharmaceutical manufacturing, pharmacovigilance, and medical affairs.
Backed by a team of over 100 skilled professionals, cutting-edge technologies, and deep collaborations with researchers and industry partners, we transform scientific innovations into tangible patient outcomes.
Our integrated approach covers the complete drug development spectrum, from molecule and vaccine design through preclinical research, late-phase clinical studies, safety monitoring, regulatory submissions, and global market access.
What We Do
- ✓ Cover the complete drug development spectrum, from molecule and vaccine design to preclinical research, late-phase clinical studies, safety monitoring, and regulatory submissions
- ✓ Broaden therapeutic capabilities, delivering integrated solutions across vaccines, biotherapeutics, and cutting-edge stem cell and gene therapy programs
- ✓ Leverage synergistic networks and hospital partnerships, enabling faster patient recruitment and efficient study execution
- ✓ Optimize regulatory and commercialization pathways, combining manufacturing expertise, advanced therapy proficiency, and CRO operational excellence

Our Core Services
Comprehensive solutions spanning the entire drug development lifecycle
✓ Preclinical Studies
Early-stage laboratory and animal testing to evaluate drug safety, efficacy, and pharmacological properties
✓ Clinical Studies
Full-spectrum clinical trial management from feasibility to final submission with regulatory expertise
✓ Stem Cell & Gene Therapy
Advanced therapy studies with specialized infrastructure, expert teams, and global regulatory alignment
✓ Biopharmaceuticals: Commercial & Manufacturing
Development and manufacturing of WHO-prequalified vaccines and biotherapeutic products
✓ Pharmacovigilance
Comprehensive drug safety monitoring, signal detection, and risk management throughout product lifecycle
✓ Medical Affairs
Scientific liaison, medical education, and real-world evidence integration for optimal outcomes
Our Mission, Vision & Goal
Driving innovation, advancing patient outcomes, and creating sustainable value
Mission
To accelerate high-impact therapies via advanced technology, optimized trials, and strategic partnerships by delivering scalable value for patients and shareholders.
Vision
To emerge as a global catalyst in clinical innovation, driving superior patient outcomes and sustainable value creation through technology-enabled, partnership-driven research excellence.
Goal
To advance healthier lives for all through transformative and translational research that bridges the gap between innovation and patient care.
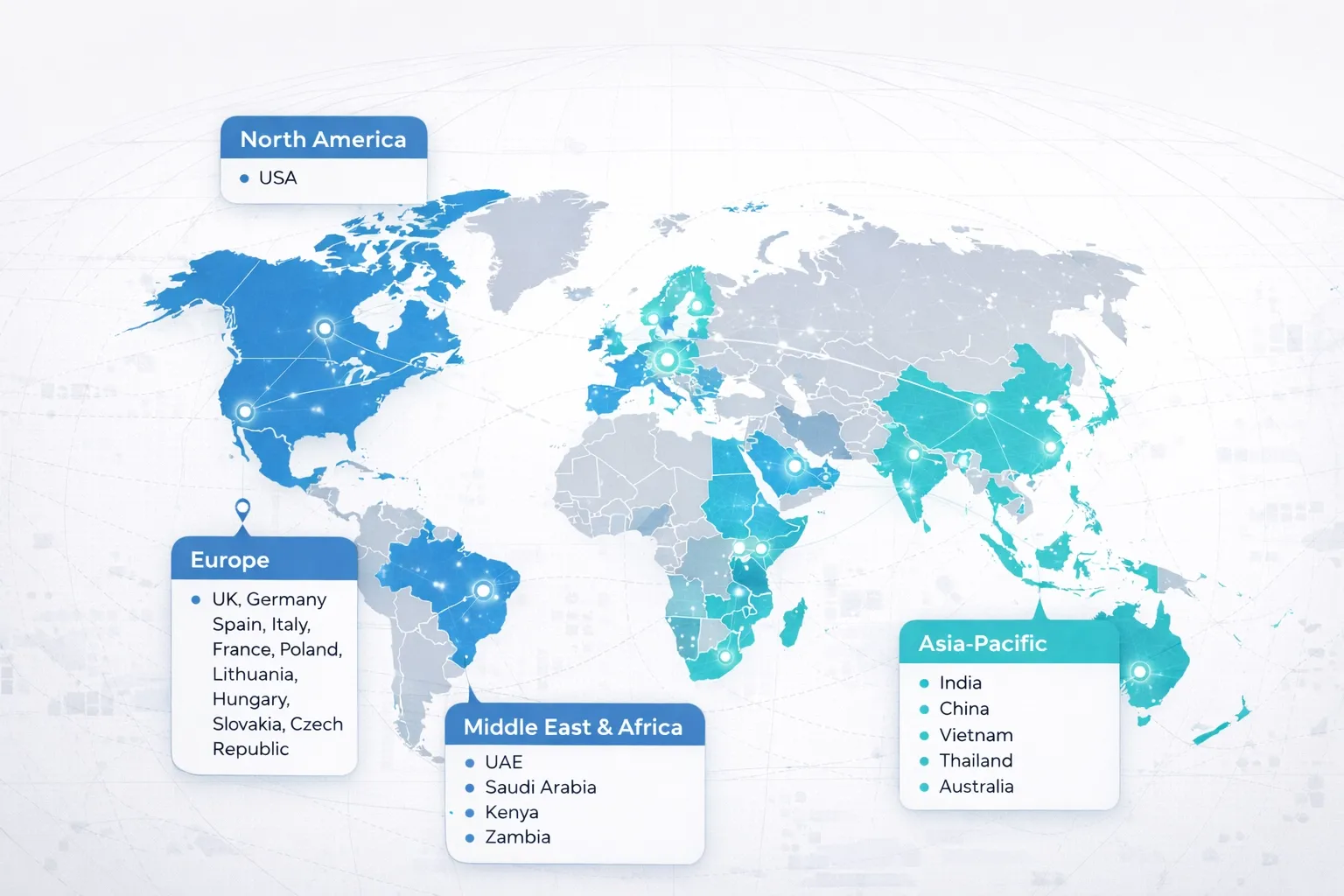
Our Global Reach
Strategic partnerships and presence across continents
Worldwide Network
Our global strategic partnerships span multiple continents, empowering us to deliver integrated solutions and regional expertise worldwide.
Our Presence in:
Europe
UK, Germany, Spain, Italy, France, Poland, Lithuania, Hungary, Slovakia, Czech Republic
Middle East & Africa
UAE, Saudi Arabia, Kenya, Uganda, Zambia
Asia-Pacific
India, China, Vietnam, Thailand, Australia
North America
USA and strategic partnerships
This global network enables us to understand diverse regulatory environments, access specialized expertise, and deliver culturally aligned solutions in every market we serve.
Leadership & Advisors
Experienced professionals driving innovation and excellence

Dr. Anit Singh
Founder & CEO
Dr. Anit Singh is a visionary biopharmaceutical executive with over two decades of global leadership spanning Medical Affairs, Clinical Research, Pharmacovigilance, Stem Cell and Gene Therapy, and Regulatory Strategy. An MBBS and MD from AIIMS, New Delhi, he combines deep scientific expertise with strategic and commercial acumen to accelerate innovation, ensure compliance, and drive strong organizational performance.
Having led over 200 clinical trials and authored 50+ scientific publications, Dr. Singh has partnered with global institutions such as WHO, ICMR, DBT, CSIR, the Bill & Melinda Gates Foundation, and the European Commission on high-impact health initiatives.
As Founder & CEO of Clinical Research Network India and Provaxa International (UAE), he is recognized for his ability to scale operations across emerging markets, build high-performing teams, and deliver sustainable growth in complex biopharma ecosystems.

Dr. Preston Izulla
Advisor
Dr. Preston Izulla is a seasoned public health leader with over 19 years of experience across Sub-Saharan Africa, partnering with global organizations such as Gavi, the Global Fund, CDC, and the Bill & Melinda Gates Foundation. Holding an MPH from Moi University, a Diploma in Vaccinology from Institut Pasteur, and an MBChB from the University of Nairobi, he combines clinical expertise with strategic vision.
Dr. Izulla has led high-impact programs in HIV/TB, maternal and child health, and vaccine safety—advancing health systems, accountability, and innovation in global vaccine research and delivery.

Dr. Sutee Yoskan
Advisor
Dr. Sutee Yoskan is an eminent vaccine scientist and public health leader from Mahidol University, Thailand, renowned for his pioneering work in dengue vaccine research and development. With extensive experience in immunology, virology, and translational vaccine science, he has played a pivotal role in advancing dengue prevention strategies across Southeast Asia.
Dr. Yoskan has collaborated with global health agencies, academic institutions, and vaccine developers to bridge research, policy, and implementation—driving impactful innovation in vector-borne disease control. His leadership and scientific vision continue to shape regional and global vaccine programs, particularly within emerging infectious disease preparedness and immunization strategy development.
100+ Skilled Professionals
Our growing team of over 100 talented professionals brings diverse expertise in clinical research, regulatory affairs, manufacturing, and medical affairs, united by a shared commitment to advancing patient care globally.
Our Strengths
What sets us apart in the medical research landscape
Integrated End-to-End Solutions
Complete drug development lifecycle coverage from preclinical through commercial access, reducing friction and accelerating time-to-market.
Global Regulatory Expertise
Deep knowledge of CDSCO, EMA, FDA, and emerging market regulations enabling seamless multi-regional submissions.
Strategic Hospital Partnerships
Network of top clinical facilities enabling faster patient recruitment, efficient study execution, and real-world evidence generation.
Advanced Technology Infrastructure
State-of-the-art facilities, GMP-compliant manufacturing, and cutting-edge laboratory capabilities supporting innovation.
Patient-Centric Approach
Commitment to improving patient outcomes through rigorous research, safety monitoring, and real-world evidence integration.
Proven Track Record
Extensive experience backed by successful regulatory approvals, global partnerships, and satisfied pharmaceutical sponsors.
Let's Accelerate Your Next Breakthrough
Partner with Provaxa to transform your research into tangible patient outcomes.
Provaxa is proudly powered by WordPress